The publication of this report led the IUPAC Commission on Nomenclature of Organic Chemistry to consider the preparation of a set of IUPAC Rules for Carbohydrate Nomenclature. This work was continued, and a revised version was endorsed in 1963 by the American Chemical Society and by the Chemical Society in Britain and published. A joint British-American committee was therefore set up, and in 1952 it published Rules for Carbohydrate Nomenclature. 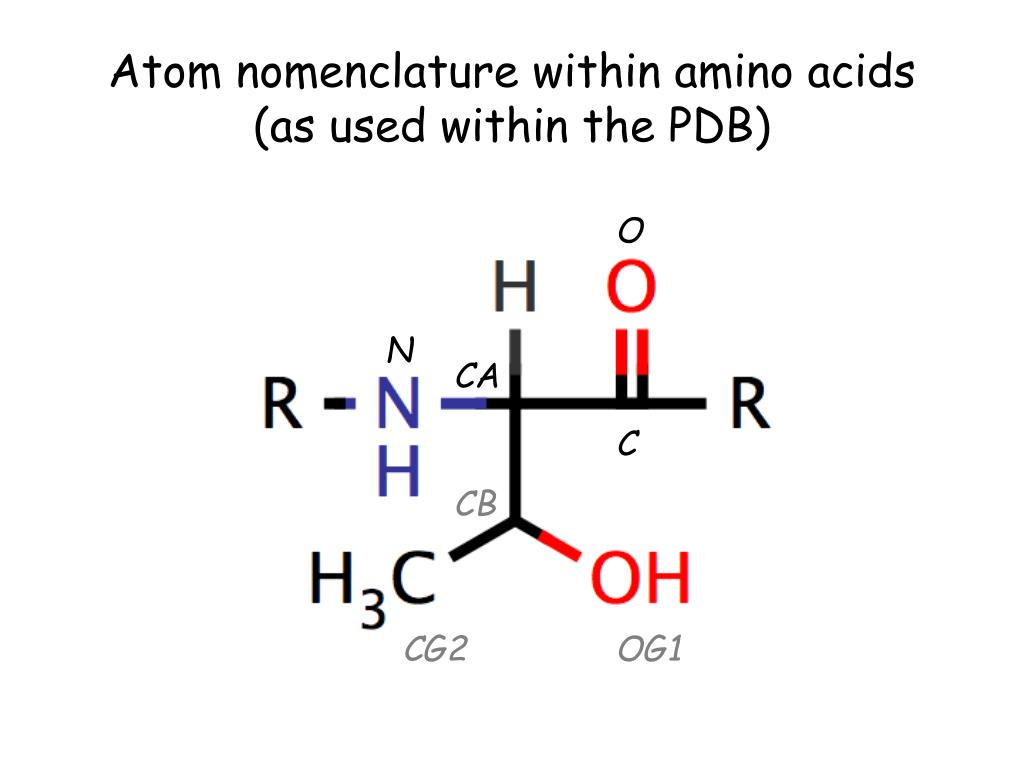
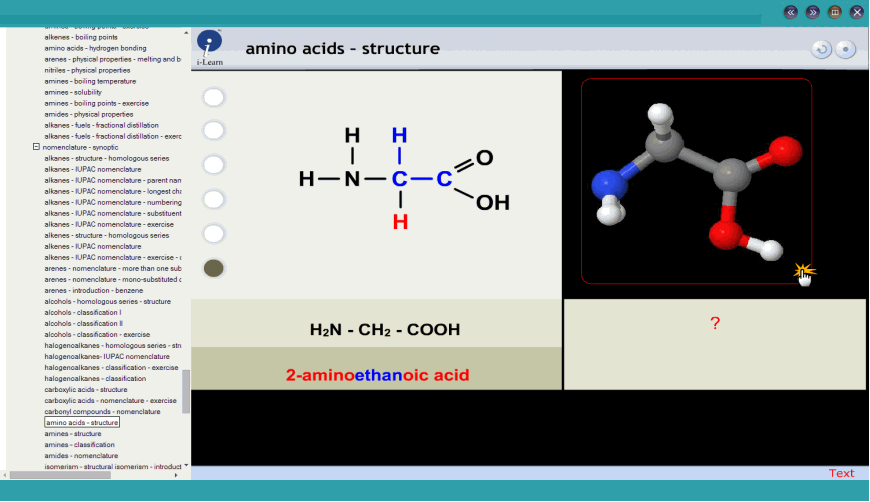
Not all problems were solved, however, and different usages were encountered on the two sides of the Atlantic. 387 and in Eur.J.Biochem., 1984,138, 9, encourage the retention of trivial names for the common a- amino acids, but systematic names are relatively straightforward thus, L-alanine is 2S-aminopropanoic add and L-histidine is 2S-amino-3-(imidazol-4-yl)-propanoic add (the name for the predominant tautomer). IUPAC IUB nomenclature recommendations (1983), reproduced in full in Amino Acids, Peptides, and Proteins, 1985, Vol. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
